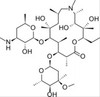
Azithromycin impurity I, EvoPure 3'-N-demethylazithromycin) is one of several impurities found in Azithromycin and can be used as a reference standard for impurity profiling.
We also offer:
- Azithromycin Impurity E, EvoPure (A074)
- Azithromycin Impurity F, EvoPure (A075)
- Azithromycin Impurity G, EvoPure (A083)
- Azithromycin impurity J, EvoPure (A082)
- Azithromycin impurity L, EvoPure (A078)
EvoPure products have been fully characterized by spectral analysis and are shipped with a comprehensive certificate of analysis containing lot-specific HPLC, MS, HNMR, and FTIR data.
| Application | Azithromycin impurity I is primarily used as a reference standard. Reference standards are well characterized compounds that can be used to help identify and/or quantify impurities in pharmaceutical compounds and antimicrobials. |
| Molecular Formula | C37H70N2O12 |
| References |
Gladue RP, Bright GM, Isaacson RE, Newborg MF (1989) In vitro and in vivo uptake of Azithromycin (CP-62,993) by phagocytic cells: Possible mechanism of delivery and release at sites of infection. Antimicrob. Agents Chemother. 33(3):277-282 PMID 2543276 Lovmar, M and Tenson T (2003) The Mechanism of Action of Macrolides, Lincosamides and Streptogramin B Reveals the Nascent Peptide Exit Path in the Ribosome."Journal of Molecular Microbiology 330 (5):1005-1014. PMID 12860123 Wolf K and Malinverni R (1999) Effect of Azithromycin plus Rifampin versus that of Azithromycin alone on the eradication of Chlamydia pneumoniae from lung tissue in experimental pneumonitis. Antimicrob. Agents Chemother. 43(6):1491-1493. PMID 10348778 |