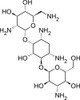
Kanamycin B Sulfate, EvoPure is a highly purified (≥99.0% pure) and fully characterized sulfate salt form of Kanamycin B. Kanamycin B is a minor component of the Kanamycin complex.
Kanamycin B is a close structural analog of DAA2 and it has an unmethylated 3’-amino group so it can be tested as a surrogate substrate for 3’-deamination.
Kanamycin is a semi-synthetic, broad-spectrum aminoglycoside antibiotic. Kanamycin was isolated from the soil bacterium Streptomyces kanamyceticus in 1957 by Hamao Umezawa at the institute of Microbial Chemistry (Tokyo). Kanamycin inhibits protein synthesis in bacteria and Mycoplasma. It is used to select for transformants (bacteria, fungi, plant, mammalian) which have been successfully transformed with a plasmid containing a Kanamycin resistance gene.
Kanamycin B Sulfate, EvoPure is freely soluble in water. It is practically insoluble in ethanol, DMSO, acetone, chloroform, ether and ethyl acetate.
We also offer:
- Kanamycin Sulfate (K008)
- Kanamycin Acid Sulfate, BP (K004)
- Kanamycin Acid Sulfate, EP (K029)
- Kanamycin A Sulfate, EvoPure (K013)
Kanamycin EvoPure products are available as highly purified fractions. Each compound is fully characterized by spectral analysis and comes with a comprehensive certificate of analysis that includes HPLC, MS, HNMR and FTIR data. These compounds can be used to study the effects of a specific Kanamycin fraction.
| Mechanism of Action | Aminoglycosides target the 30S ribosomal subunit, lodging between the 16S rRNA and S12 protein. This interferes with the translational initiation complex causing misreading of the mRNA, and production of a faulty or nonexistent protein. |
| Spectrum | Kanamycin is broad-spectrum, however, it is mostly used against aerobic Gram-negative bacteria. |
| Microbiology Applications | Kanamycin is commonly used in clinical in vitro microbiological antimicrobial susceptibility tests (panels, discs, and MIC strips) against Gram-positive and Gram-negative microbial isolates. Medical microbiologists use AST results to recommend antibiotic treatment options for infected patients. Representative MIC values include:
Kanamycin B was used as a structural analog of DAA2, an important enzyme in the Gentamicin biosynthetic pathway, to study the use of the biosynthetic pathway of Gentamicin to gain insights into engineering safer Aminoglycoside derivatives (Huang et al, 2015). Kanamycin is one of the most widely used antibiotics yet its biosynthetic pathway remains unclear. Authors reconstructed the entire biosynthetic pathway through the expression of putative biosynthetic genes from S. kanamyceticus in the non-aminoglycoside-producing Streptomyces venezuelae, demonstrating the potential of pathway engineering to direct in vivo production of more robust aminoglycosides (Park et al, 2011). |
| Plant Biology Applications | Kanamycin is often used in Agrobacterium mediated transformation with the npt II gene as selection marker. Researchers used Kanamycin in combination with cefotaxime to control bacterial growth in tomato transformation (Kaur and Bansal, 2010). |
| Molecular Formula |
C18H37N5O10 · xH2SO4 (lot-specific) |
| Source | Semi-synthetic; Originally derived from Streptomyces kanamyceticus |
| References |
Huang et al. used Kanamycin B Sulfate, EvoPure (TOKU-E) to study the roles of oxidoreductase GenD2 and transaminase GenS2. "Delineating the biosynthesis of Gentamicin X2, the common precursor of the Gentamicin C antibiotic complex." Aragão FJL and Brasileiro ACM (2009) Positive, negative and marker-free strategies for transgenic plant selection. Braz. J. Plant Physiol. 14(1):1-10 Davis BD (1987) Mechanism of bactericidal action of Aminoglycosides. Microbiol. Rev. 51(3):341-350 Grant CCR, Konkel ME, Cieplak Jr W, and Tompkins LS (1993) Role of flagella in adherence, internalization, and translocation of Campylobacter jejuni in nonpolarized and polarized epithelial cell cultures. Infect. and Immun. 61(5):1764-1771 Kaur P and Bansal KC (2010) Efficient production of transgenic tomatoes via Agrobacterium-mediated transformation. Biologica plantarum 54 (2): 344-348 Lorz H, Baker B and Schell J (1985) Gene transfer to cereal cells mediated by protoplast transformation. Molec. And Gen. Genetics 199:178-182 Nguyen QH et al (2019) Overexpression of the GmDREB6 gene enhances proline accumulation and salt tolerance in genetically modified soybean plants. Sci Rep. 9:19663 PMID 31873128 Park JW et al (2011) Discovery of parallel pathways of Kanamycin biosynthesis allows antibiotic manipulation. Nat. Chem. Biol. 7(11):843-852 PMID 21983602 Sharma MK, Solanke AU, Jani D, Singh Y and Sharma Ak (2009) A simple and efficient Agrobacterium-mediated procedure for transformation of tomato. J. Biosci. 34(3): 423–433 Sneader W (2005) Drug Discovery: A history. John Wiley & Sons pp 302 Takahashi T, Morotomi Ma nd Nomoto K (2004) A novel mouse model of rectal cancer established by orthotopic implantation of colon cancer cells. Cancer Sci. 95(6):514-519 PMID 15182433 |
| MIC | Escherichia coli| 3.125|| Pseudomonas aeruginosa| 3.125|| |