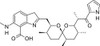
Calcimycin (syn. A23187) is a potent Ca2+ ionophore and carboxylic acid antibiotic isolated from the soil bacterium Streptomyces chartreusis (NRRL 3882) in 1974. It was isolated from the bacterial culture as a mixed calcium-magnesium salt, which can be converted to the free acid form.
Calcimycin is a spiroketal substituted by pyrollic and benzoxazolyl groups which allow its high affinity and selectivity for calcium. It also has high affinity for Mg2+ and Mn2+. It is used as a research tool for calcium regulation. A mobile ion-carrier, it forms complexes with divalent cations, thus equilibrating the inter- and intracellular concentrations. It allows Mn2+ into the cell to quench intracellular dye fluorescence and can be used in flow cytometry. It can be used with visible light-excitable indicators including calcium green, magnesium green and calcium orange and others. Calcimycin has activity against bacteria, fungi, protozoa, and Mycobacteria. Calcimycin can be used as a research tool to study the role of divalent cations in biological systems, such as increasing the levels of intracellular Ca2+ in intact cells. It can also be used for in-situ calibrations of fluorescent Ca2+ indicators.
Calcimycin is soluble in ethanol, methanol, DMSO and DMF. It is slightly soluble in water. Dissolve in in DMSO or ethanol prior to preparing low concentrations of aqueous solutions.