Three Historical Breakthroughs for Neomycin
What is Neomycin?
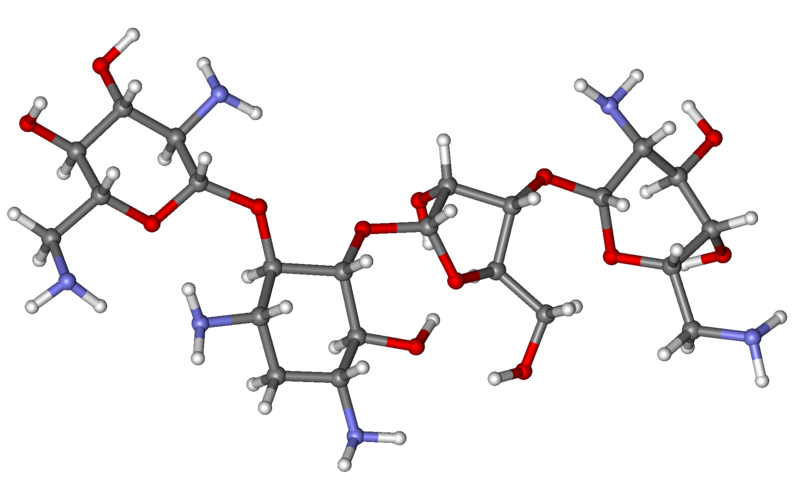
Neomycin C. Neomycin is a therapeutic agent, typically used in its sulfate form. Characteristically, neomycin sulfate is a white or yellowish-white powder, and is very soluble in water and very slightly soluble in alcohol.
Neomycin sulfate is not a chemically pure compound, and is mostly comprised the active component, neomycin B, and several related impurities. The permitted quantities of neomycin sulfate impurities are set by the United States Pharmacopoeia (USP) and European Pharmacopoeia (EP).
- Impurity A: R1 = H, R2 = NH2 : 2-deoxy-4-O-(2,6-diamino-2,6- dideoxy-α-D-glucopyranosyl)-D-streptamine (neamine or neomycin A-LP),
- Impurity B: R1 = CO-CH3, R2 = NH2 : 3-N-acetyl-2-deoxy-4-O-(2,6- diamino-2,6-dideoxy-α-D-glucopyranosyl)-D-streptamine (3-acetylneamine),
- Impurity D: R1 = H, R2 = OH: 4-O-(2-amino-2-deoxy-α-Dglucopyranosyl)-2-deoxy-D-streptamine (paromamine or neomycin D),
- Impurity E: R1 = R3 = H, R2 = CH2-NH2, R4 = OH: 4-O-(2-amino-2-deoxy-α-D-glucopyranosyl)-2-deoxy- 5-O-[3-O-(2,6-diamino-2,6-dideoxy-β-L-idopyranosyl)- β-D-ribofuranosyl]-D-streptamine (paromomycin I or neomycin E),
- Impurity F: R1 = CH2-NH2, R2 = R3 = H, R4 = OH: 4-O-(2-amino-2-deoxy-α-D-glucopyranosyl)-2- deoxy-5-O-[3-O-(2,6-diamino-2,6-dideoxy-α-Dglucopyranosyl)-β-D-ribofuranosyl]-D-streptamine (paromomycin II or neomycin F),
- Impurity G: R1 = H, R2 = CH2-NH2, R3 = CO-CH3, R4 = NH2 : 3-N-acetyl-2-deoxy-4-O-(2,6-diamino-2,6-dideoxy-α-Dglucopyranosyl)-5-O-[3-O-(2,6-diamino-2,6-dideoxy- β-L-idopyranosyl)-β-D-ribofuranosyl]-D-streptamine (neomycin B-LP).
According to the USP and EP, a max of 2% of impurity A and 3% - 15% of impurity C is allowed in pharmaceutical grade neomycin sulfate. Furthermore, there cannot be more than 5% of any other impurity, totaling to no more than 15% total impurities.
Common Applications
Neomycin is routinely used in cell culture, microbiology, and plant biology as a selection agent to isolate genetically transformed cells and tissues. In clinical settings, neomycin is used as a topical agent. Due to its extreme nephrotoxicity, neomycin is not administered intravenously.
History of Neomycin
The history of neomycin dates back to 1949 when is was discovered in the lab of Selman Waksman at Rutgers University.

Produced by Streptomyces fradiae through a fermentation process, neomycin is nephrotoxic to humans, and some individuals may have allergic reactions to it.
Neomycin Discoveries
Neomycin C is 50% as active as Neomycin B Against Staphylococcus epidermidis
Conducted as an in vitro study at The Upjohn Company in Kalamazoo, Michigan, researchers set out to determine the different responses of neomycin C and B to Staphylococcus epidermidis. Initially, Kiyoshi Tsuji had measured that neomycin C's response against Staphylococcus aurerus was 1/3 that of neomycin B. Following tests of Staphylococcus epidermidis, Tsuji discovered that the 1/3 rule didn't apply. He was able to apply a response factor of 50% (as compared to neomycin B) to neomycin C. This is now the factor that is used when calculating the microbiological equivalency of potencies determined by gas chromatography.
The GLC assay method is a valuable tool for quantifying neomycin fractions and monitoring its biosynthesis and degradation processes
In a "Comparative Study of Responses to Neomycins B and C by Microbiological and Gas-Liquid Chromatographic Assay Methods" members from the Upjohn Company in Kalamazoo, Michigan tested the different assay methods that are capable of quantifying neomycins B and C. Historically, there are several chemical methods that are unfortunately time-consuming and not applicable in a lab environment that tends to quantify a large number of neomycin products on a regular basis. However, Kiyoshi Tsuji and John Robertson from The Upjohn Company developed a gas-liquid-chromatographic (GLC) method, which has the ability of separating individual neomycin isomers. Additionally, the GLC method allows for quantification of neomycins C and B at a greater facility than the alternative methods, which are known as the agar-diffusion method and the turbidimetric method. Tsuji and Robertson set up a test to compare the responses of neomycins B and C via the GLC and the other traditional methods to determine the results. In the end, the GLC method results correlated well with the value obtained by the microbiological agar-diffusion assay method. It is mentioned that the importance and merit of the GLC method is that it has the capability of separating and quantifying neomycins B and C in approximately 30 minutes.
A 4-minute process has been developed to separate and detect three neomycin components
Under optimized conditions, scientists have developed a simple method to separate neomycin components through a capillary electrophoresis, without the need of a derivatization step. Given the quick pace at which this process operates, it could provide a new alternative to the current processes that both the US and European Pharmacopoeia use on a daily basis. An experiment was conducted to further test the validity of this 4-minute process which revealed promising results. Using a 25kV as the optimized separation voltage, scientists determined that the separation time of all the neomycin complexes was less than 4 minutes. Additionally, the test has high simplicity and repeatability factors which may make this test a viable alternative method for scientists and researchers around the world.
The Research Potential of Neomycin
The composition of neomycin is generally overlooked in genetic transfection studies as well as its use in a clinical environment. The individual neomycin components and impurities do not appear to be extensively studied which makes it difficult to fully appreciate the consequences of batch-to-batch variability of allowed quantities of neomycin impurities. Taking a closer look at the individual fractions and impurities of neomycin will allow us to better understand this valuable antibiotic, and may pave a path to defining more stringent impurity specifications which may potentially diminish the allergenic and nephrotoxic properties of neomycin.
Resources:
1Neomycin sulfate. Drug standard query database. N.p., n.d. Web. 14 Mar. 2017. http://www.drugfuture.com/standard/search.aspx
2Robertson, J. H., Baas, R., Yeager, R. L., & Tsuji, K. (1971). Antimicrobial Activity of Neomycin C Against Staphylococcus epidermidis. Applied Microbiology, 22(6), 1164-1165. Retrieved March 09, 2017
3Tsuji, K., Robertson, J. H., Baas, R., & MCinnis, D. J. (1969). Comparative Study of Responses to Neomycins B and C by Microbiological and Gas-Liquid Chromatographic Assay Methods. Applied Microbiology, 18(3), 396-398. Retrieved March 09, 2017
