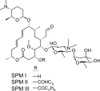
Spiramycin is a broad-spectrum 16-membered ring macrolide antibiotic composed of a mixture of spiramycin I, II, and III. Spiramycin was first isolated by PINNERT-SINDICO in 1954 from Streptomyces ambofaciens.
Spiramycin is a bacterial protein synthesis inhibitor, it works by irreversibly binding to the P-site on the 50s ribosome, preventing peptide bond formation and translocation. Spiramycin is effective against gram-negative and gram-positive bacteria, as well as some Toxoplasma parasites.
Spiramycin has shown anti-obesity effects. In a recent study (2016), it was shown that spiramycin inhibits adipogenesis in 3T3-L1 cells and ameliorates obesity and associated metabolic indications in HFD-fed mice.
Synonyms: Rovamycine, Formacidine, Foromacidine, NSC 55926, Kitasamycin
We also offer:
- Spiramycin I (S094)
| Mechanism of Action | Macrolide antibiotics, like spiramycin, inhibit bacterial growth bacteriostatically by targeting the 50S ribosomal subunit preventing peptide bond formation and translocation during protein synthesis. Resistance to spiramycin is commonly attributed to mutations in 50S rRNA preventing spiramycin binding allowing the cell to synthesize proteins free of error. |
| Spectrum | Spiramycin is a broad-spectrum antibiotic targeting a wide range of gram-positive and gram-negative bacteria. In addition, spiramycin has demonstrated activity against Toxoplasma parasites. |
| Microbiology Applications | Spiramycin is commonly used in clinical in vitro microbiological antimicrobial susceptibility tests (panels, discs, and MIC strips) against gram positive and gramnegative microbial isolates. Medical microbiologists use AST results to recommend antibiotic treatment options for infected patients. Representative MIC values include:
|
| Eukaryotic Cell Culture Applications | Spiramycin has shown various anti-obesity effects in 3T3-L1 preadipocytes and in mice models. Spiramycin is able to inhibit adipogenesis in 3T3-L1 preadipocytes, and reduce lipid accumulation in-vitro. Spiramycin is also able to decrease adipogenic gene expression by reducing the expression of adipogenic transcriptional factors like, PPARγ, SREBP1c, and C/EBPα, in preadipocytes. Spiramycin increases AMPK activity during early adipogensis in 3T3-L1 preadipocytes. Spiramycin ameliorates HFD-induced obesity and obesity-related parameters in C57BL/6 mice. |
| Molecular Formula | Spiramycin I: C43H74N2O14 |
| Elemental Analysis | Composition: Spiramycin I: Not less than 80.0% Spiramycin II: Not more than 5.0% Spiramycin III: Not more than 10.0% Sum of Spiramycin I, II, III: Not less than 90.0% |
| References |
Lovmar, Martin, and Tanel Tenson. "The Mechanism of Action of Macrolides, Lincosamides and Streptogramin B Reveals the Nascent Peptide Exit Path in the Ribosome."Journal of Molecular Microbiology 330.5 (2003): 1005-014. |
| MIC | Actinomyces odontolyticus| 2.67|| Alcaligenes faecalis| 80|| Bacillus subtilis| 5|| Capnocytophaga gingivalis| 64|| Clostridium spiroforme| 0.12 - 256|| Eikenella corrodens| 2|| Enterococcus| 0.25 - ≥512|| Enterococcus faecalis| 10|| Escherichia coli| 80|| Fusobacterium nucleatum| ≥64|| Micrococcus luteus| 2.5|| Mycobacterium smegmatis| 10|| Mycoplasma fermentans| 2 - 4|| Mycoplasma genitalium| 0.12 - 1|| Mycoplasma hominis| 32 - >64|| Mycoplasma penetrans| 16|| Mycoplasma pneumonia| ≤0.015 - 0.25|| Pasteurella haemolytica| 128|| Peptostreptococcus micros| 64|| Porphyromonas asaccharolytica| 16|| Porphyromonas gingivalis| 9.2|| Prevotella intermedia| 16|| Prevotella melaninogenica| 3|| Pseudomonas aeruginosa| >80|| Salmonella typhimurium| >80|| Staphylococcus aureus| 2.5 - 80|| Streptococcus| 0.025 - 64|| Streptococcus pneumonia| ≤0.031 - >64|| Streptococcus pyogenes| ≤0.031 - >64|| Treponema hyodysenteriae | >100|| Ureaplasma urealyticum| 4 - 32|| Veillonella parvula | 64|| |