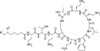
Polymyxin B Sulfate, USP is a polypeptide antibiotic mixture composed of Polymyxins B1, B2, and B3 fractions, with B1 and B2 fractions comprising the majority of the mixture. Polymyxin B components are structurally identical with the exception of a variable fatty acid group on each fraction. Results from in vitro studies have shown marginal differences in MIC data when comparing the fractions. Polymyxin B Sulfate is freely soluble in aqueous solution.
Polymyxin B Sulfate, USP conforms to United States Pharmacopoeia specifications.
We also offer:
- Polymyxin B1 Sulfate, EvoPure (P037)
- Polymyxin B1-I Sulfate, EvoPure (P038)
- Polymyxin B2 Sulfate, EvoPure (P039)
- Polymyxin B3 Sulfate, EvoPure (P040)
- Polymyxin B6 Sulfate, EvoPure (P054)
- Polymyxin E1 Sulfate, EvoPure (P055)
- Polymyxin E2 Sulfate, EvoPure (P056)
- Polymyxin B ReadyMadeTM Solution (P174)
EvoPure products are purified single antibiotic fractions, most are >99% pure. Highly pure EvoPure Polymyxin products can be used to analyze the specifc effects of individual Polymyxin B fractions.
| Mechanism of Action | Polymyxin B targets and alters the permeability of lipopolysaccharide (LPS) found in Gram-negative bacteria leading to lysing of the cell. Polymyxin B only needs to interact with LPS, it is not required to enter the cell. |
| Spectrum | Polymyxin B Sulfate targets the outer membrane of Gram-negative bacteria especially Pseudomonas aeruginosa. |
| Impurity Profile |
Polymyxin B1||4135-11-9|C56H98N16O13|1204| Polymyxin B2|||C55H96N16O13|1190| Polymyxin B3|||C55H96N16O13|1190| Polymyxin B1-I|||C56H98N16O13|1204| Composition of Polymyxins: Polymyxin B3: Not more than 6.0% Organic Impurities |
| Microbiology Applications | Polymyxin B Sulfate is commonly used in clinical in vitro microbiological antimicrobial susceptibility tests (panels, discs, and MIC strips) against Gram-negative microbial isolates. Medical microbiologists use AST results to recommend antibiotic treatment options. Representative MIC values include:
Media Supplements:Polymyxin B is routinely used as a selection agent in several types of isolation media: Perfringens agar - Supplement A and Supplement B Perfringens Agar - SFP and TSC Selective Supplements Columbia Blood Agar - Campylobacter Selective Supplement (Skirrow) Columbia Blood Agar - Campylobacter Selective Supplement (Blaser-Wang) Brucella medium - Brucella Selective Supplement MYP Agar - Polymyxin B Bacillus Selection Supplement Legionella CYE Agar - Legionella BMPA-α Selective Supplement Legionella CYE Agar - Legionella MWY Selective Supplement Campylobacter Agar - Campylobacter Selective Supplement (Preston) PALCAM Agar - PALCAM Selective Supplement Legionella CYE Agar - Legionella GVPC Selective Supplement m-CP Medium - Membrane C. perfringens Selective Supplement Burkholderia cepacia Agar Base - Burkholderia cepacia Selective Supplement ORSAB - ORSAB Selective Supplement Campylobacter Agar Base - Modified Preston Campylobacter Selective Supplement Brucella Medium Base - Modified Brucella Selective Supplement Legionella CYE Agar - Legionella GVPN Selective Supplement ChromogenicListeria Agar - Chromogenic Listeria Selective Supplement ChromogenicBacillus cereus Agar - Chromogenic Bacillus cereus Selective Supplement ChromogenicListeria Agar - Chromogenic Listeria Differential Supplement Schneider et al. found that the combination of the CFTR drugs (cystic fibrosis transmembrane conductance regulator) and Polymyxin B showed synergistic in vitro activity against P. aeruginosa. this pathogen causes infection in those with cystic fibrosis (CF). Authors They also investigated this antimicrobial mode of action, and found the bacterial outer membrane damage to be unique from the effect of each individual compound. This is an exciting new angle on the treatment of polymyxin-resistant lung infections (Schneider et al, 2016). |
| Plant Biology Applications | Polymyxin B Sulfate was successfully tested to counteract phytopathogenic Gram-negative bacterial growth including different strains of Pseudomonas viridiflava and Erwinia carotovora. Polymixin B Sulfate was shown to reduce bacterial growth of different strains of Pseudomonas viridiflava at low concentrations, (0.08 µg/ml) and Erwinia carotovora growth at slightly higher concentrations (0.25 µg/ml) (Selim et al. 2005). Polymyxin B has been shown to elict alkaloid accumulation in E. californica. Treatment at 0.04 mg/ml for 4 hours showed a 5.5x increase in Jasmonate levels. |
| References |
Kassamali et al used Polymyxin B1, Polymyxin B2, Polymyxin B3, and Polymyxin B1-I (TOKU-E) to test for synergistic and antagonistic effects against various Gram-negative organisms in: "Microbiological assessment of Polymyxin B components tested alone and in combination" Crass et al used Polymyxin B1 and B2 (TOKU-E) to investigate the pharmacokinetics of Polymyxin B in: Pharmacokinetics of Polymyxin B in hospitalized adults with cystic fibrosis. Cao G et al (2009) Development and validation of a reversed-phase high-performance liquid chromatography assay for Polymyxin B in human plasma. J. Antimicrob. Chemother. 62(5):1009-1014 PMID 18765414 Mueller MJ, Brodschelm W, Spannagl E and Zenk MH (1993) Signaling in the elicitation process is mediated through the octadecanoid pathway leading to jasmonic acid. Proc. Natl. Acad. Sci. USA 90(16):7490-7494 PMID 11607420 Newton BA (1956) The properties and mode of action of the Polymyxins. Bacter. Rev. 20(1):14-27 PMID 13303920 Orwa JA et al (2001) Isolation and structural characterization of Polymyxin B components. J Chromatogr A 912(2):369-373 PMID 11330807 Schneider E et al (2026) An 'unlikely' pair: The antimicrobial synergy of Polymyxin B in combination with the systic fibrosis transmembrane conductance regulator drugs KALYDECO and ORKAMBI. ACS Infect. Dis. 2016, 2, 7, 478–488 Selim S, Negrel J, Govaerts C, Gianinazzi S and Tuinen van D (2005) Isolation and partial characterization of antagonistic peptides produced by Paenibacillus sp. strain B2 isolated from the sorghum Mycorrhizosphere. Appl. Environ. Microbiol. 71(11):6501–6507 PMID 16269674 Tam VH, Cao H, Ledesma, KR, Hu M (2011) In vitro potency of various Polymyxin B components.55(9):4490-491 PMID 21709096 Zavascki AP, Goldani LA, Li J and Nation RL (2007) Polymyxin B for the treatment of multidrug-resistant pathogens: A critical review. J. Antimicrob. Chemother. 60(6):1206-1215 PMID 17878146 |
| MIC | Diplococcus pneumoniae| ≥400|| Fusobacterium necrophorum| 8.1 - 100|| Haemophilus influenzae| ≥0.8|| Pseudomonas aeruginosa| 0.25 - 1|| |