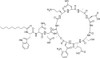
Daptomycin is a unique lipopeptide antibiotic derived from Streptomyces roseosporus that is effective against Gram-positive bacteria. This holoenzyme is composed of three subunits, encoded by the dptA, dptBC, and dptD genes, each responsible for incorporating particular amino acids into the peptide. The cloned dpt gene cluster allows study of Daptomycin biosynthesis and discovery of novel lipopetides through pathway engineering.
Daptomycin is soluble in water, DMSO, methanol, and ethanol.
We also offer:
- Daptomycin ReadyMadeTM Solution (D108)
| Application | Daptomycin is the only commercially available lipopeptide antibiotic. The Daptomycin gene cluster has been cloned and sequenced (McHenney et al, 1998). By exchanging heterologous peptide synthetase modules into the coding region, it should be possible to produce novel derivatives of Daptomycin. These can then undergo chemical modification to develop candidates for evaluation. |
| Mechanism of Action | Daptomycin peptides interact with anionic phospholipids in the membrane of Gram-positive bacteria. Peptides integrate into the membrane by a calcium-mediated process and interact with membrane phospholipid head groups and acyl chains. Finally, a pore or channel is formed which can kill the cell by membrane disruption/depolarization or cellular uptake of Daptomycin leading to disruption of intracellular processes such as synthesis of DNA, RNA, and protein which results in bacterial cell death. The activity of Daptomycin is dependent on the presence of physiologic levels of free calcium ions. Researchers found a gradual dissipation of membrane potential, thus membrane depolarization is an inherent part of its mechanism of action (Silverman et al, 2003). |
| Spectrum | Daptomycin targets Gram-positive bacteria including GRE (glycopeptide-resistant Enterococci) and MRSA (methicillin-resistant Staphylococcus aureus). |
| Microbiology Applications | Daptomycin has been used to study the impact of Staphylococcal accessory regulator (sarA) on Daptomycin susceptibility of Staphylococcus aureus. The mutation of sarA did not affect the Daptomycin MIC but resulted in increased susceptibility in vivo in the context of an established biofilm (Weiss et al, 2009) Representative MIC ranges for Daptomycin from the Antimicrobial Index appear below.
|
| Molecular Formula | C72H101N17O26 |
| Solubility | Soluble in water (100 mg/mL), DMSO (100 mM), methanol, and ethanol (50 mM). |
| References | Baltz RH, Miao V and Wrigley SK (2005) Natural products to drugs: Daptomycin and related lipopeptide antibiotics. Nat. Prod. Rep. 22(6):717-741 PMID 16311632 McHenney MA Hosted TJ, Dehoff BS, Rosteck PR and Baltz RH (1998) Molecular cloning and physical mapping of the Daptomycin gene cluster from Streptomyces roseosporus. J. Bacteriol. 180(1):143-151 Miao V et al (2006) Genetic engineering in Streptomyces roseosporus to produce hybrid lipopeptide antibiotics. Chem. Biol. 13(3):269-76 PMID 16638532Silverman JA, Perlmutter NG, and Shapiro HM (2003) Correlation of Daptomycin bactericidal activity and membrane depolarization in Staphylococcus aureus. Antimicrob. Agents Chemother. 47(8):2538-44 PMID 12878516 Straus, SK and Hancock REW (2006) Mode of action of the new antibiotic for Gram-positive pathogens Daptomycin: Comparison with cationic antimicrobial peptides and lipopeptides. Biomem. 1758(9):1215-1223 Weiss EC et al (2009) Impact of sarA on Daptomycin susceptibility of Staphylococcus aureus biofilms in vivo. Antimicrobial. Agents. Chemother. 53(10):4096-4102 PMID 19651914 |